It’s not uncommon to see people advocating for more screening in health care. Sometimes they mean screening for diseases (like major depression), sometimes they mean risk factors (like consuming alcohol), and sometimes they mean situations (like ‘intimate partner violence’).
The original underlying assumption for disease screening was that diseases arise in healthy people and then inevitably progress through somewhat predictable stages, with increasing pathological abnormalities and danger along the way. Intuitively, earlier recognition (or increased recognition) of diseases should therefore lead to earlier treatment, improved outcomes, and reduced overall costs. It seems logical! The earlier the better!
Screening for disease is a very old idea. We’ve had a lot of opportunity to test the theory! (The idea that screening works for risk factors and life situations is a newer idea, and less well tested).
Back in 1861 the British physician Horace Dobell proposed that a routine check-up (involving an exhaustive history, physical examination, and battery of laboratory tests, leaving no stone unturned) was “the only means by which to reach the evil and to obtain the good.”
The concept was more explicitly linked to cancer in the early 1900’s, when Dr. Charles Childe claimed1 that “…if every case of cancer came under the notice of the [physician] at the earliest possible moment...it requires no stretch of the imagination...to say that the majority...would be cured.”
In the 1920’s, the American Medical Association officially endorsed the universal periodic health exam. In their view, “medical experience of the benefits of periodic examinations of presumably healthy persons is sufficiently widespread to make any detailed reference superfluous”2, with another author3 claiming that “The proposal that healthy people be examined at regular intervals… is notable for the logic of its theory and the slowness of its adoption.” In other words, it was a ‘no brainer’, and the science was settled. No need to argue, just get on with it! Sound familiar?
Building on that foundation, in the subsequent decades (and up to the present day), the American Society for the Control of Cancer, later to become the American Cancer Society, focused on the importance vigilance for the early warning signs of cancer, while heavily promoting screening procedures including Pap smears, breast exams, mammograms, PSA testing, etc.
By the early 1970s, however, there was some concern about the value of universal periodic health exams, in part because it had become apparent that most people who felt well were, in fact, reasonably healthy. You had to wade through a lot of healthy people to find the sick ones, and that’s a lot of work if it involves an exhaustive history, physical examination, and battery of laboratory tests! It’s not an efficient way to detect serious disease. Many of the abnormalities you do find are inconsequential (i.e. bunions, hemorrhoids, etc.)4
Since then, the focus has gradually shifted to the periodic and targeted use of proven disease screening modalities, as suggested by organizations including the Canadian Task Force on Preventive Health Care, the U.S. Preventive Services Task Force, and others. In theory, these organizations are objective, principle driven, methodical, and evidence based.
So, rather than simply examining every body part and system in detail, the current approach is to limit the search, based on specific criteria and good quality evidence. By this standard, the list of proven screening modalities has become pretty limited. Even so, there’s widespread faith in the magical power of screening, with advocates constantly pushing for more.
It’s a complex topic, hard for anyone to understand, so intuition often triumphs over actual data. Studies show that doctors and patients alike overestimate the benefits and minimize the harms. By times, even the organizations that produce the screening guidelines have fallen prey to political influence, including the idea that screening works as well for risk factors and living situations as it does for disease.
It’s a mess, in other words! Let’s pick over the entrails, to see if we can find meaning in the chaos.
Screening, in principle
The object of screening for disease is to discover those among the apparently well who are in fact suffering from disease.
In theory… screening is an admirable method of combating disease, since it should help detect it in its early stages and enable it to be treated adequately before it obtains a firm hold on the community.
In practice, there are snags.
(Principles and Practice of Screening for Disease, 1968)
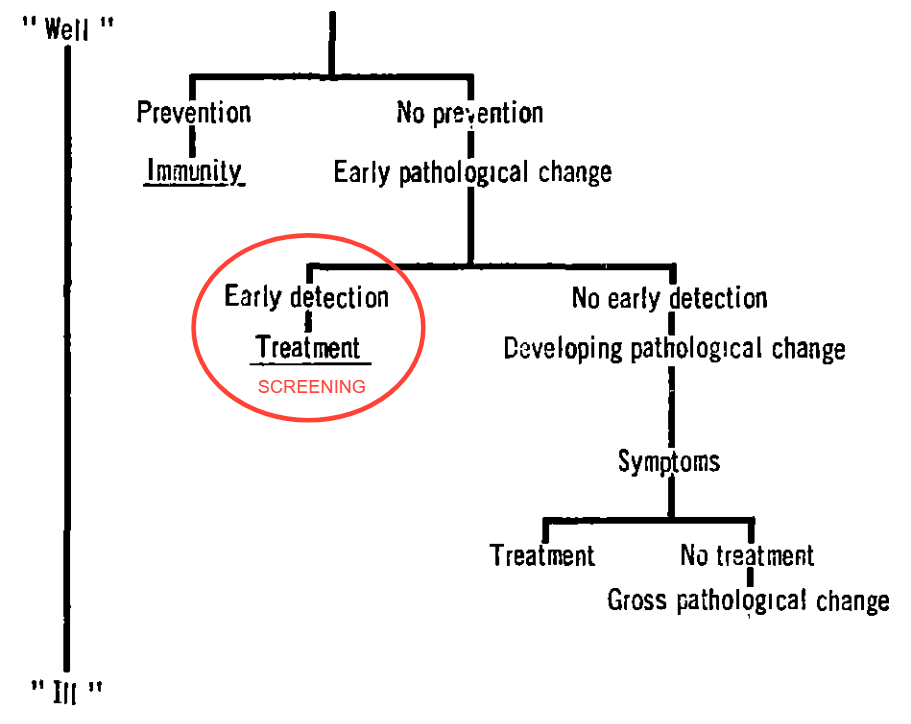
In the late 1960’s, the World Health Organization (WHO) published Public Health Paper #34, Principles and Practice of Screening for Disease, written by Wilson and Jungner.
According to the WHO, “the purpose of Public Health Papers is to stimulate international thinking, discussion, and planning by the publication of the personal ideas, observations, and suggestions of individuals or groups”. Obviously, the science of screening wasn’t settled!
Wilson and Jungner laid out the basic premise of disease screening, using the following diagram.
In screening, we seek to intervene EARLY, in the sweet spot after the disease has produced pathological changes (something that can be identified through physical examination, blood tests, x-rays, etc.) and before symptoms arise. Screening can’t work when there’s nothing yet detectable with the available tools, and it’s too late for screening when symptoms develop, which is presumably when the patient would seek medical care, per usual.
Wilson and Jungner also defined the basic principles of screening, which have held up well over the years. In a 2018 systematic review of the topic, Dobrow et al reviewed the original WHO document and 40 subsequent publications, concluding that “…Wilson and Jungner’s principles of screening were ahead of their time, [while] our review showed a shift in subsequent sets of screening principles toward more operational and implementation issues.” In other words, the underlying theory hasn’t really changed, but with practice we’ve learned more about the practical administrative details.
So, let’s look at those principles.
The condition sought should be an important health problem.
This one seems obvious.
The importance of the problem is considered from the point of view of both the individual and the community.
A rare but devastating disease may be worth screening for, if the cost or effort of screening is relatively low and there is effective treatment available. Phenylketonuria, for example, is a rare (1-in-22,000 births) condition easily detectable by screening all newborns. For those affected, a modified diet begun as soon as possible after birth and continued for life can prevent intellectual disability, seizures, behavioral problems, and mental disorders, resulting in normal health and a normal life span.
Common but milder treatable conditions may also be worth screening for, if they have serious consequences for individuals or the community when not discovered and treated. For example, in communities where tuberculosis is endemic, screening can identify infected individuals early, allowing for their treatment before serious complications arise, while also reducing transmission to others.
Facilities for diagnosis and treatment should be available.
Again, sort of obvious.
A screening test is almost never diagnostic. It simply splits the screened population into two groups, those more likely to have the disease (screen positive), and those less likely to have it (screen negative).
Your screening program is designed to turn up previously unrecognized possible cases of disease, so the screen positive group will need timely access to further testing to confirm (or refute) the diagnosis, with treatment to follow.5
The cost of case-finding (including diagnosis and treatment of patients diagnosed) should be economically balanced in relation to possible expenditure on medical care as a whole.
Whether you are screening for rare but individually devasting disease or a common but individually mild disease, you need to consider the relative costs of screening versus not screening.
Screening programs require money, staff, space, and equipment, all of which are limited. You don’t want to expend valuable resources screening for diseases that are incredibly rare6, completely trivial7, or untreatable. You also don’t want to spend added money on early detection if early treatment (prompted by screening) is no better or no cheaper than late treatment (prompted by symptoms).
The natural history of the condition, including development from latent to declared disease, should be adequately understood. In particular, there should be a recognizable latent or asymptomatic stage.
This one takes a bit of explaining. It’s critically important.
I’ve explained in previous posts8 how diseases are defined and why it’s important to understand the natural history.
In screening, you are looking for a specific group, the patients who have a disease but don’t yet know that they have it. That requires a period in the natural history of the condition during which symptoms are either absent or minimal. If worrisome symptoms develop early in the course of an illness, then patients will seek care early because they have symptoms. These patients already get care early, and they probably won’t benefit from earlier detection. They require diagnosis, not screening.
Also, the asymptomatic or minimally symptomatic phase has to be long enough to allow for the development of some sort of pathological changes detectable with a screening test.9 Rapidly progressive conditions don’t lend themselves to screening, even if they start out asymptomatic, in part because there are practical limits to how often you can screen, and in part because earlier detection may not make much difference in a rapidly growing malignancy.10
So, we set screening intervals based on the natural history of the disease, knowing when pathological changes develop, how fast they progress, when they are detectable by testing, and how long it is likely to be before symptoms appear.11
Rather than screening everybody, it helps to have a target population. If a subset of the population is at greater risk, then the screening can be targeted, increasing its effectiveness while reducing costs. We only screen for cervical cancer, for example, in those who actually have a cervix and have been sexually active, starting at an age when the disease has had sufficient time to develop.
Let’s look at a hypothetical disease, Cancer Q, which starts on Day 1 with an abnormality in a single cell deep within an organ. That abnormal cell slowly divides over the ensuing months to the point where, left untreated, Cancer Q would cause death on Day 365. A Cancer Q tumor would be visible on an x-ray on Day 90, with symptoms developing on Day 180. This gap between Day 90 and Day 180 is the ‘latent phase’. Treatment in the patients who present with symptoms is 50% effective (half still die on Day 365, the other 50% are cured). Sometimes, Cancer Q is fortuitously detected between Day 90 and Day 180, when the patient is having an x-ray for some other reason and the radiologist happens to notice the tumor. In these cases, when Cancer Q is detected between Day 90 (tumor detectable on x-ray) and day 180 (symptoms develop), there’s 90% cure rate. On the face of it, early detection of Cancer Q using x-rays sounds like a good idea, because earlier treatment improves the chances of a cure.
However, there are other principles to consider.
There should be a suitable screening test or examination which is available, accessible and acceptable to the population. Screening should be a continuing process, not a "once and for all" project.
In screening, what you want is a quick way to sort out which patients are more likely to have the disease (screen positive), and those less likely to have it (screen negative).
As Dobrow et al put it (emphasis added), “Screening test performance should be appropriate for the purpose, with all key components specific to the test… being accurate (e.g., in terms of sensitivity, specificity and positive predictive value) and reliable or reproducible. The test should be acceptable to the target population, and it should be possible to perform or administer it safely, affordably and efficiently…. Screening test results should be clearly interpretable and determinate (e.g., with known distribution of test values and well-defined and agreed cut-off points) to allow identification of the screening participants who should (and should not) be offered diagnostic testing and other post screening care.”
So, assuming we want to screen for Cancer Q, we would have to do x-ray screening on ALL of the patients at risk for cancer Q (which means we know the risk factors), in that 90-day window when x-rays would show the tumor and symptoms have not yet developed. Since there’s a steady stream of new Cancer Q patients in the pipeline, we end up screening every at-risk patient with an x-ray once every three months, which could be complicated and expensive, not to mention the risks associated with all those x-rays.
We would have to accept that Cancer Q patients wouldn’t have any visible x-ray changes before Day 90, so those cancers wouldn’t be detected. We call these ‘false negatives’. A test with too many false negatives is considered less ‘sensitive’. A highly sensitive test is preferred.12 People lose faith in screening when they are told they don’t have a disease and later find out that they actually do have it! They expect the result to be ‘determinate’ - either you have the disease or you don’t!
On the other hand, you want some assurances that the x-ray abnormalities seen in Cancer Q are easily identifiable (no ‘borderline’ or ‘indeterminate’ results!), somewhat unique to Cancer Q, and unlikely to appear in other less serious but more common diseases, otherwise you’ll get a lot of people with spots on their x-ray that aren’t cancer Q. We call these “false positives”. A test with too many false positives is considered less ‘specific’. A highly specific test is preferred, because sorting out borderline results and false positives can be complicated, expensive, and procedurally risky (bleeding and infections with biopsies, for example), not to mention the anxiety caused by telling people who thought they were healthy that they might have a disease.13
Unfortunately, there’s usually a tradeoff between sensitivity, specificity, and cost. It’s rare to find a test that’s highly specific, highly sensitive, and cheap!
Some tests are less ‘acceptable’ than others. Any screening test which is highly invasive, inconvenient, or unpleasant will have a reduced chance of success, because many individuals will simply refuse the test. In screening for colon cancer, for example, colonoscopy is invasive, unpleasant, and somewhat risky. The alternative is testing first for occult blood in the stool, but some patients still find that collecting and submitting a stool sample is inconvenient and unpleasant. Mammograms involve inconvenient travel to a screening centre and then having your breasts unpleasantly crushed, so some patients decline.
All that being said, some cancers do have a long latent phase, and we have identified screening tests that balance sensitivity, specificity, and acceptability. Cancer of the cervix, for example, has a long latent phase, allowing for Pap Smear screening at regular intervals. Those who found the Pap Smear to be invasive and unpleasant will be pleased to know that now (or soon) they can self-test for HPV at home.
There should be an accepted treatment for patients with recognized disease. Importantly, early treatment must have a greater benefit than late treatment, and there should be an agreed policy on whom to treat.
This one also seems obvious, but there are nuances.
Any treatment, intervention, and follow-up care triggered by screening must modify the natural history of the disease, improving outcomes (e.g. increased functioning or quality of life, decreased disease-specific mortality, etc.).
Clearly, there’s no point in the earlier detection of an incurable disease unless, for some reason, your patient wants more time to ‘get their affairs in order’ before their inevitable death.
Furthermore, even when there is an effective treatment, if the outcome is the same regardless of when in the course of the disease the person receives therapy, then there is no justification for earlier diagnosis.
If our hypothetical Cancer Q carried the same 50% cure rate regardless of whether it was diagnosed early (by x-ray) or late (based on symptoms) then early diagnosis would have no benefit.
On the other hand, if Cancer Q treatment initiated after Day 180 is only 50% curative, whereas treatment initiated between Day 90 (tumor detectable on x-ray) and day 180 (symptoms develop) has a 90% cure rate, then earlier diagnosis is beneficial!
However, this is not always the case, and it’s easy to be misled.
Imagine two patients with Cancer Q:
Patient #1 develops symptoms (sometime on or after Day 180), seeks care, gets a diagnosis, and is treated, but unfortunately still dies on Day 365. They had up to 6 months before death in which they knew there was a problem, and they spent a good chunk of it ‘battling cancer’, getting treatment which made them unwell while proving to be futile.
Patient #2 is asymptomatic but is fortuitously found to have cancer Q by x-ray (sometime on or after Day 90 and before Day 180). They too are diagnosed and treated, and they too succumb on Day 365. They had up to 9 months before death in which they knew there was a problem, and they also spent a good chunk of it ‘battling cancer’.
On the face of it, it looks like Patient #2 benefitted from the earlier diagnosis, because they lived longer after the diagnosis was made. In fact, both patients died as expected, when expected, with neither one benefitting from their treatment.
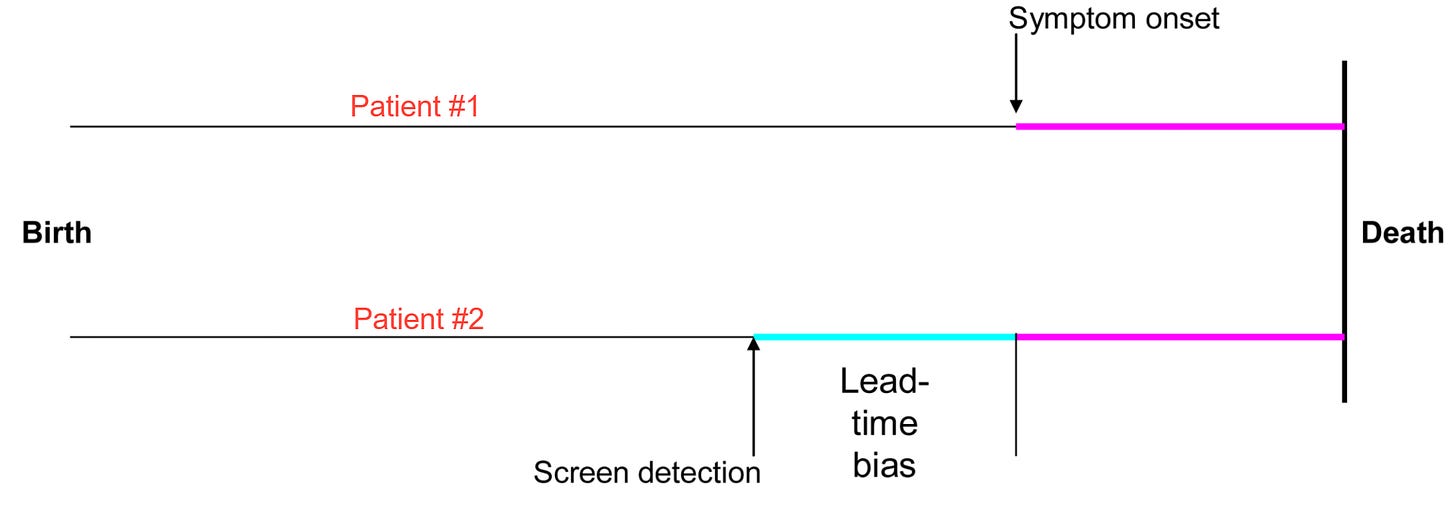
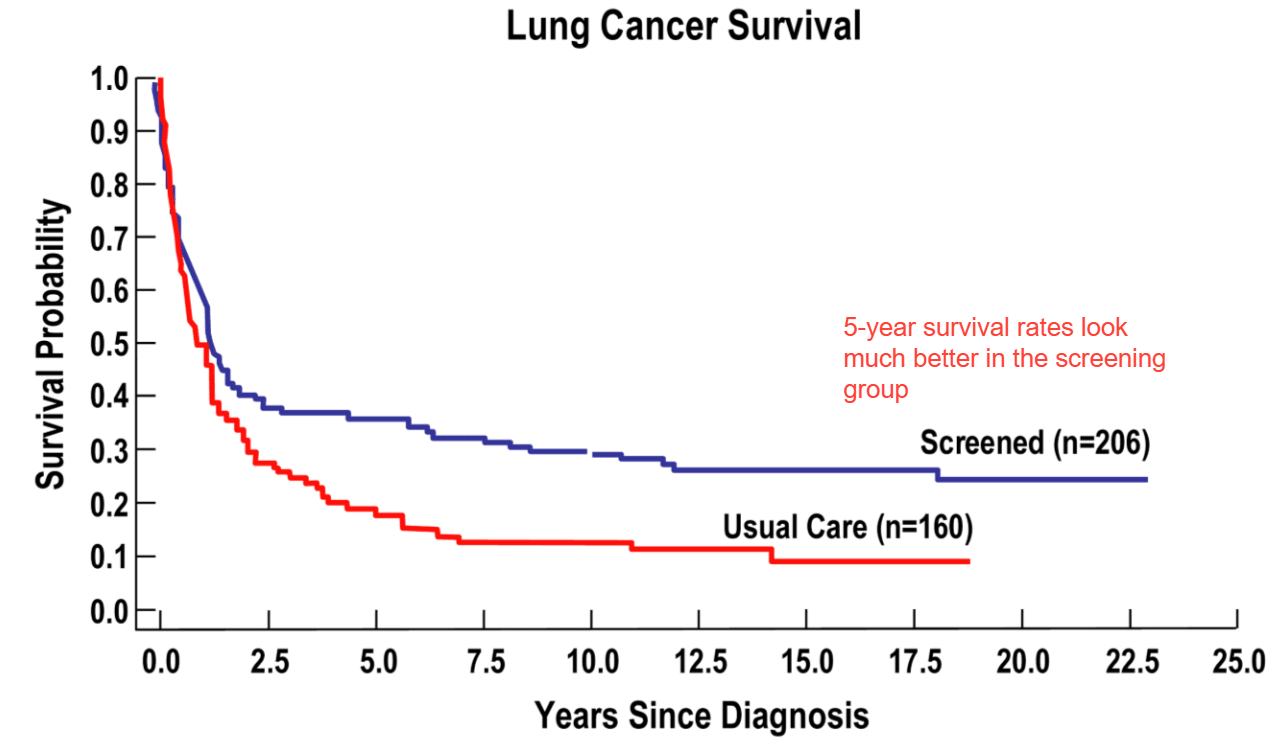
This is called ‘lead time bias’. Treatment hasn’t altered the natural history of the disease, but it looks as though it has, because with earlier diagnosis you live longer than ‘usual’ (or ‘expected’) after the diagnosis is made. This explains why 5-year survival rates cannot and should not be used to judge the value of cancer screening, although they often are. Screening, simply by detecting disease earlier, will inevitably improve 5-year survival without necessarily saving lives.14
At the same time, any negative effects of knowing about the disease (anxiety, depression, family upheaval, treatment effects and side-effects, economic consequences, etc.) will kick in when the diagnosis is made, which means that Patient #2 may actually have suffered more, in the final analysis.
And this brings us back to the importance of knowing the natural history of the disease. In the case of Cancer Q (or any cancer), we don’t know what happens before the x-ray (or other test) shows changes or the symptoms appear. Furthermore, every time we discover a way to detect the disease earlier, we have to relearn the natural history.
“… screening alters the hitherto understood natural history of a given form of cancer. A useful analogy is picturing cancer as an iceberg of disease. Symptomatic lesions comprise the tip of iceberg - the part that is above the waterline - and, as such, can be easily observed. However, cancer includes a wide array of heterogeneous lesions that vary substantially in behavior. In the absence of screening, these asymptomatic lesions lie below the waterline of observation and little or nothing is known about the natural history of such tumors. Therefore, the information that has been gathered about the expected course and prognosis of a given cancer type is only based upon the behavior of symptomatic lesions. A new screening modality essentially dips beneath the water surface for the first time and reveals previously hidden lesions. This complicates accurate predictions regarding the course of screen-detected disease, because the natural history of this early lesion has not previously been observed and followed. These asymptomatic lesions may be far less aggressive in nature or may follow a different disease course than those diagnosed on the basis of symptoms; one cannot simply assume that the natural history of screen-detected lesions would follow the same course as that of symptomatic tumors.” (Croswell et al., 2010, p. 4)
So, my previous example of Cancer Q was greatly oversimplified, because it assumed that every case of cancer Q proceeded from onset to death through an identical stepwise progression of increasingly abnormal stages. In real life, tumor biology varies from patient to patient, and cancer is simply an umbrella term for many different disease processes, some of which look the same under the microscope but behave quite differently.
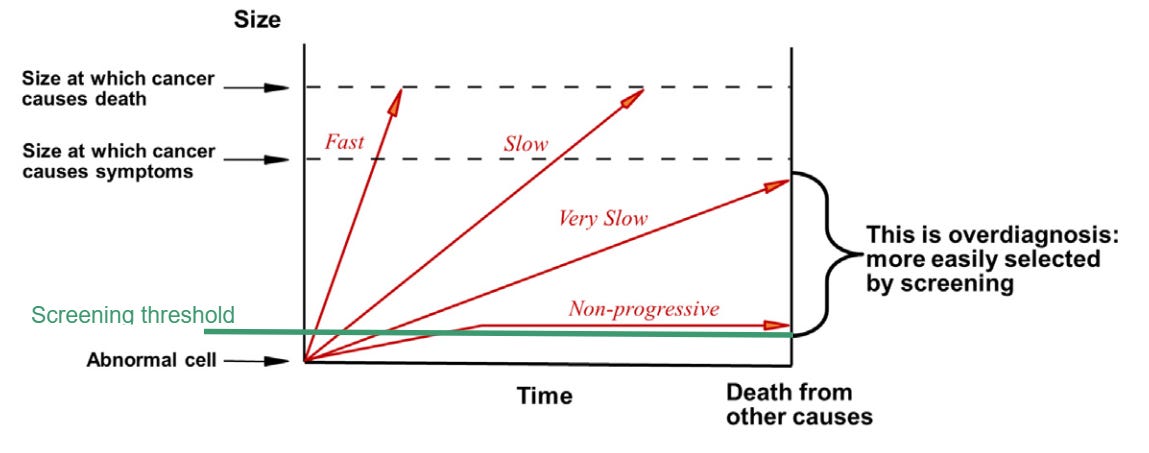
As the diagram suggests, some tumors are indolent (non-progressive), some grow very slowly, some grow less slowly, and others are rapidly progressive. Others, not shown, spontaneously regress without any treatment.15
So, what we don’t know, when we find a tumor of a given size (whether by screening or by diagnostic testing), is how long it has been there and how fast it is growing. In the diagram above, where the green line indicates the smallest lesion detectable by screening, a lesion that size could be:
just barely large enough to be detected, but never going to grow any larger, never going to cause symptoms, and never going to cause death (non-progressive).
large enough to be detected, but very slow growing, such that the patient would likely die of other causes long before any symptoms appeared (very slow).
large enough to be detected and growing slowly enough that there’s a benefit to detection before symptoms appear, thereby averting (or delaying) death due to this cancer (slow).
large enough to be detected but growing rapidly enough that there’s only a brief window of opportunity before symptoms appear, in which case it may not be possible to avert (or delay) death due to this cancer (fast).
When early diagnosis followed by early treatment actually improves patient outcomes, as with the slow growing tumors in the above graph, then there should be an eventual decline in the number of patients with advanced disease and the number of deaths. An effective early detection strategy ‘pulls’ advanced cancers out of the future and treats them now, when they are less advanced, resulting in an increase in the number of early-stage cancers now, with a corresponding decrease in late-stage disease (and death) later.
However, in the non-progressive and very slow cases (i.e. the cases that would never have been diagnosed without screening), early detection and treatment exposes the patient to an unhelpful diagnosis followed by harmful treatment. This is overdiagnosis. In these situations, you see more early-stage tumors and no decline in late-stage disease and death.
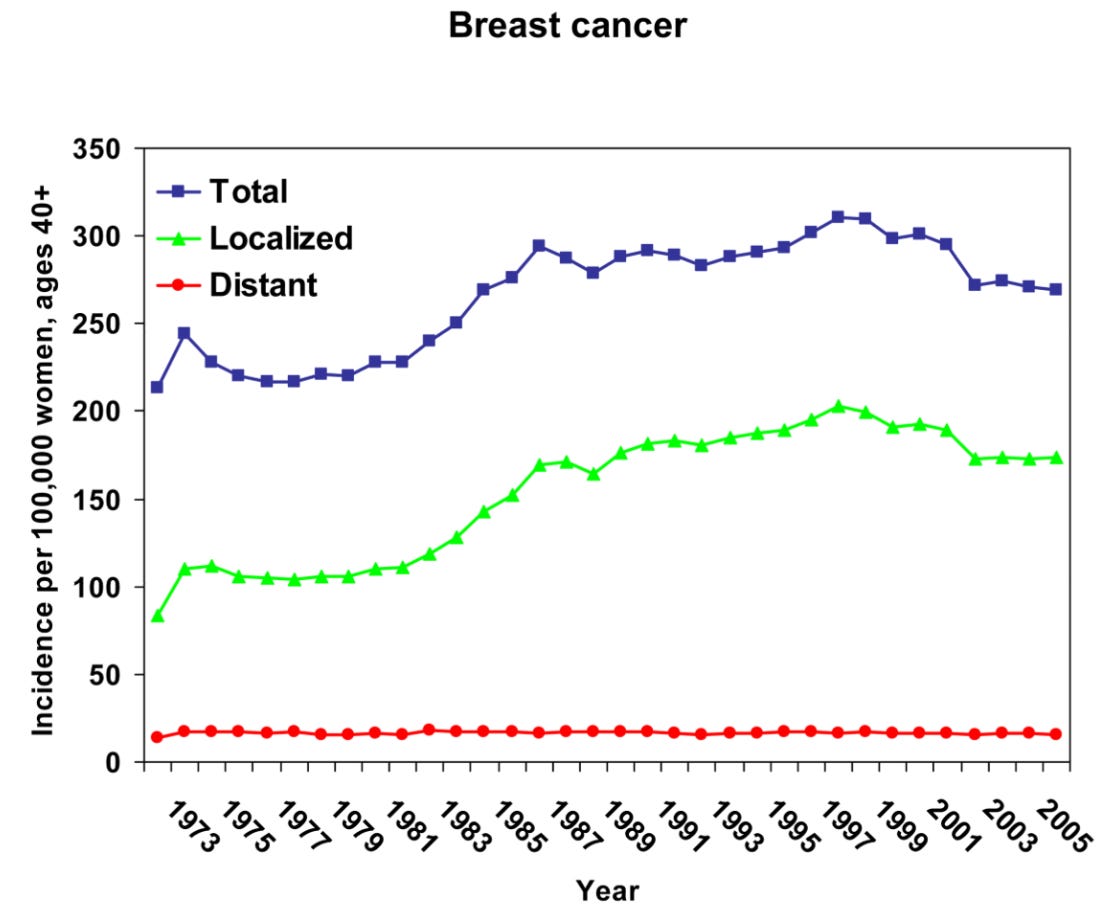
That overdiagnosis occurs is not surprising, because cancer screening tests are always going to be more effective at identifying the slower growing lesions. For every tumor, there’s an interval in which it is asymptomatic but detectable by the screening test. This window of opportunity is shorter for the rapidly growing lesions and longer for the slowly growing lesions. With slow-growing lesions, therefore, there’s a greater likelihood that the screening test will be done during the window of opportunity. This is called ‘length-biased sampling’.
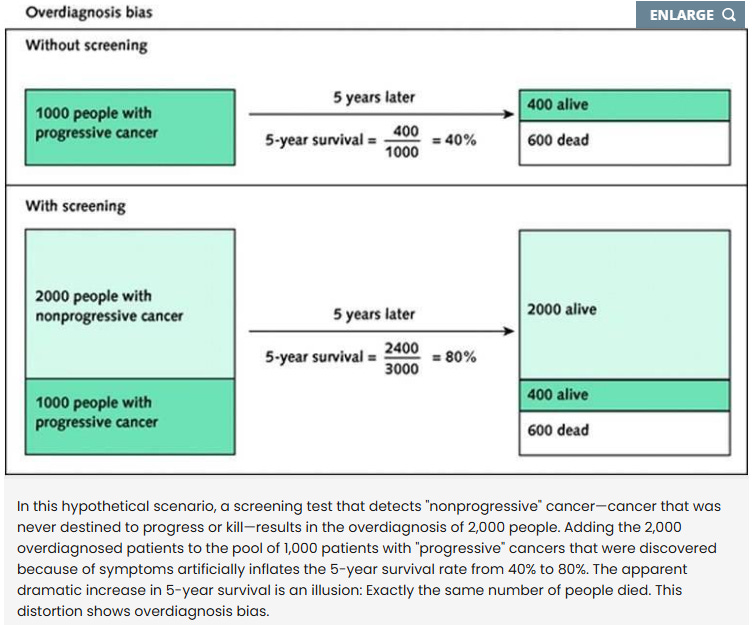
Screening tests, by design, turn up more cancers. Some will be the ones we are looking for (early stage, more easily treatable, with a better prognosis), but some will be the ones that don’t need to be treated at all. We should learn how to tell the difference, but that requires knowing the natural history of the disease!
Because the patients with screen-detected disease don’t necessarily have the same prognosis as those with symptomatic disease, their treatment plans will probably be different. We need to know what treatments work best in each group. Every time we’ve exposed a new part of the disease iceberg, we need to study the newly exposed bit, because it’s something we haven’t seen before.
“This matter resolves itself into two questions: (1) Does treatment at the pre-symptomatic border-line stage of a disease affect its course and prognosis? (2) Does treatment of the developed clinical condition at an earlier stage than normal affect its course and prognosis?”
“...without well-planned surveys, carried out in advance of the main body of medical opinion, the view that early diagnosis and treatment successfully improves the outlook for the condition in question is likely to become generally accepted. This in turn automatically renders unethical planned randomized trials of intervention by treatment, following early diagnosis; with the result that ideas about the effect of treatment pass into the realm of folklore rather than that of scientific knowledge.”
(Wilson and Jungner, p28)
Too often, we jump in and start treating the screen-detected ‘early’ disease exactly the same as we’ve always treated the symptomatic ‘late’ disease, and then we never really know whether we’ve improved the prognosis. In doing so, we’ve set a new ‘standard of care’ with no supporting evidence. From that moment on, it’s hard to reset patient expectations. It’s even harder to do research, because any research that doesn’t compare a new approach to ‘usual care’ is considered unethical.
This was the case with prostate cancer, for example. As PSA screening became more common in the 1980s, prostate cancer diagnoses rose dramatically, particularly the low-risk tumors. The initial inclination was to treat those cancers in the usual way with surgery, radiation, etc., causing the inevitable side-effects and complications. However, screening had unearthed a huge reservoir of latent disease, the sort that was indolent or slow-growing and unlikely to cause symptoms or prove fatal16. These patients were more likely to be harmed by the side effects of treatment than the disease itself. Now, the preferred approach is to monitor for a worsening of the disease, so-called ‘watchful waiting’. Even so, based on the historical overdiagnosis and overtreatment, it remains difficult to convince every patient that they can safely ‘live with cancer’. Some would still rather ‘get it out of there’, risks be damned. As for screening, current guidelines generally recommend against any prostate cancer screening in men 70 or older (or with a life expectancy of less than 10 years), as any newly diagnosed prostate cancer is unlikely to impact their natural lifespan.
In summary…
Screening works, but only where there’s a long enough latent phase, a reasonable test that detects the problem before symptoms appear, AND early treatment that makes a measurable difference to the eventual outcome. Various learned groups are constantly looking at the principles and the evidence and offering their sage advice as to which conditions should be screened for. It’s a fairly short list, but there’s constant pressure to make it longer. It’s hard to counter the hype. There isn’t a week goes by without somebody somewhere suggesting that something new should be screened for, or that we should add a new (generally more expensive) method to screen for an old disease.
For you as an individual, no matter whether your screening test was negative or positive, you’ll still think it was a good thing, even if the screening itself was totally unnecessary! Negative screen? Great, you don’t have cancer! False positive? Yes, it was nerve-wracking when the first test said there might be a problem, but you’re glad all those subsequent tests showed that you’re free and clear! True positive? Well, it’s great to know you found out about it early, so now it’s cured!
At the same time, the evidence-based screening guidelines are widely resisted by patients and physicians, in part because it seems patently obvious that earlier recognition of diseases will lead to earlier treatment, improved outcomes, and reduced overall costs. People are oblivious to the harms.
For example, women generally understand that false positives do happen with screening mammography, and of course they are reassured to find out that the suspicious abnormality on their mammogram turned out to be nothing. Far fewer are aware of the fact that screening can detect actual cancers that might never have progressed, i.e. the indolent or slow-growing ones. They are glad that the mammogram turned up a cancer and that it was then ‘cured’, even when it might never have caused problems. They exaggerate the benefits of mammography and ignore or minimize the harms.
Ultimately, there are ways to address these issues, but it won’t be easy. Powerful forces keep pushing in the other direction.
In future posts, I’ll look at specific examples where screening has been applied or suggested, and I’ll consider how we might do better in the future.
In the meantime, you might find it interesting to read the following…
Childe, CP. The Control of a Scourge, or How Cancer is Curable. New York: E.P. Dutton & Company; 1907.
Emerson H. Periodic medical examinations of apparently healthy persons. JAMA 1923;80:13761381.
Edie EB. Health Examinations Past and Present and Their Promotion in Pennsylvania. Am J Pub Health 15:602, 1925.
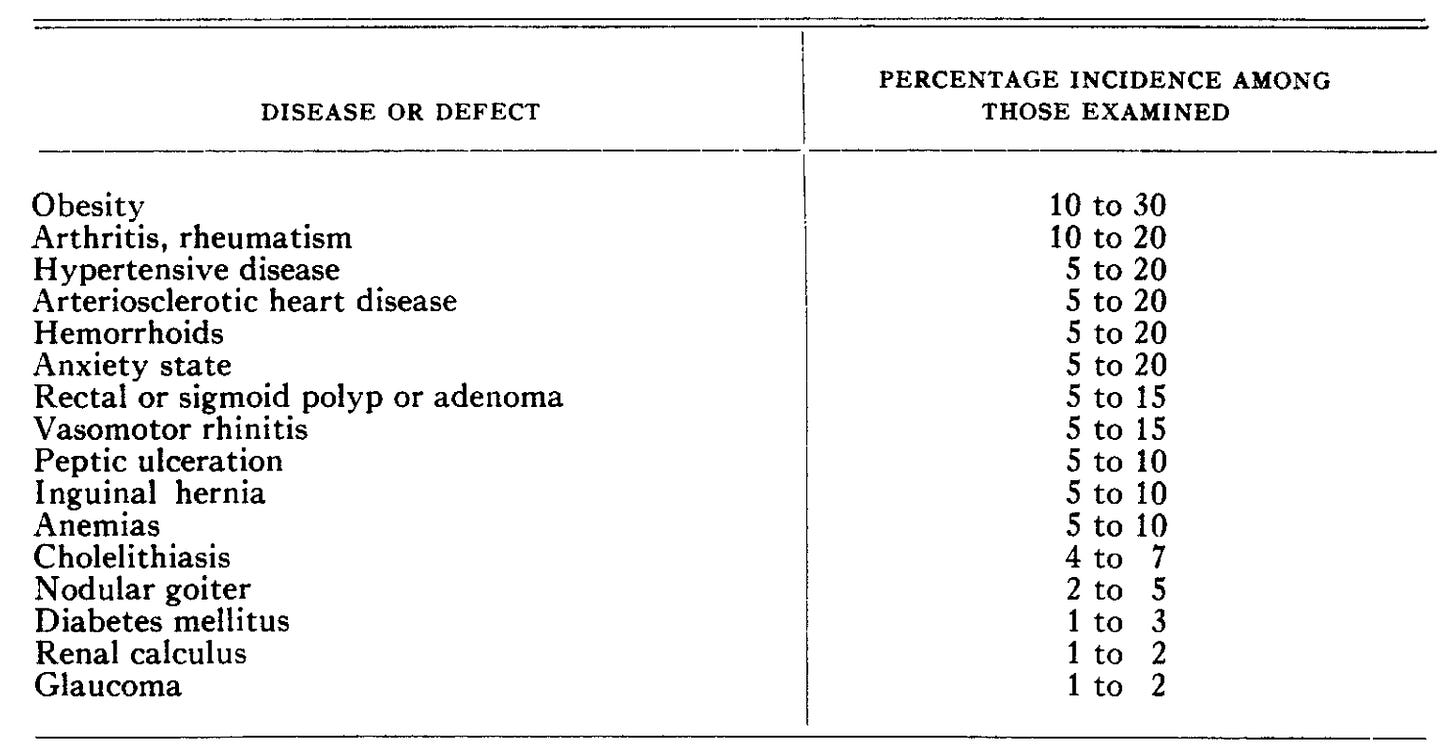
A publication from 1959, touting the value and limitations of the periodic health exam, included the following table listing the sorts to things to be found. Some are relatively trivial.
Seems obvious, but I once spent over a year as a member of a committee planning for a new colon cancer screening program, based on at risk citizens being provided sampling kits to test their bowel movements for ‘occult blood’ (meaning blood not visible to the naked eye). Patients testing positive would need further investigation by colonoscopy. It was simple enough to mail out the kits and process the samples. The biggest challenges involved ensuring that we had adequate facilities and personnel to do the required diagnostic colonoscopies in a timely fashion.
Another example from my past life in administration. There was a group lobbying for screening for sickle cell anemia in the native Nova Scotia black population (the group who have been here since the province was settled). It’s a small population, and it’s known that the sickle cell gene is extremely rare in that group, because there’s only one pediatric hospital in the province, they see every child with sickle cell anemia, and they do the genetic testing on the relatives when a case is identified. Screening wasn’t going to add any useful information, although it was hard to convince the lobbyists that the decision was based on data, not discrimination.
At the risk of being controversial, asymptomatic Covid might fall in the trivial category, given that there’s no symptoms, no treatment, and generally no consequences. At the height of the pandemic, however, some thought that the asymptomatic patients should self-isolate, to prevent transmission. That would only work if you could test everybody, every day (or even every hour) - anything less would still leave an asymptomatic but infectious group roaming the streets!
Of note, all screening tests have a threshold below which they cannot reliably detect a lesion, which means that some screen-negative patients will still have very early disease not detectable using current screening technology. Mammograms, for example, are unreliable for detecting very small breast cancer lesions, as are low-dose chest CT examinations for lung cancer. In other words, your mammogram might miss the fact that you actually do have breast cancer (a false negative).
Cancer of the pancreas might be an example.
Screening intervals can be modified as we learn more, or when new screening methods are discovered. This explains why Pap smears are done less frequently now than they were in the past, and also why they are being replaced by HPV testing, recognizing that the Human Papilloma Virus is generally present and detectable before the cervical cells show changes detectable by Pap smear.
It’s been in the news lately that mammograms are less sensitive in patients with ‘dense’ breasts. Because breast density falls more or less on a bell curve, 50% of women have dense breasts. The proposed solution for these women is to do breast MRI scans, which adds new challenges of affordability, complexity, cost, etc.
PSA blood test for prostate cancer, for example, are overly sensitive and nonspecific, leading to a lot of unnecessary investigations and treatment.
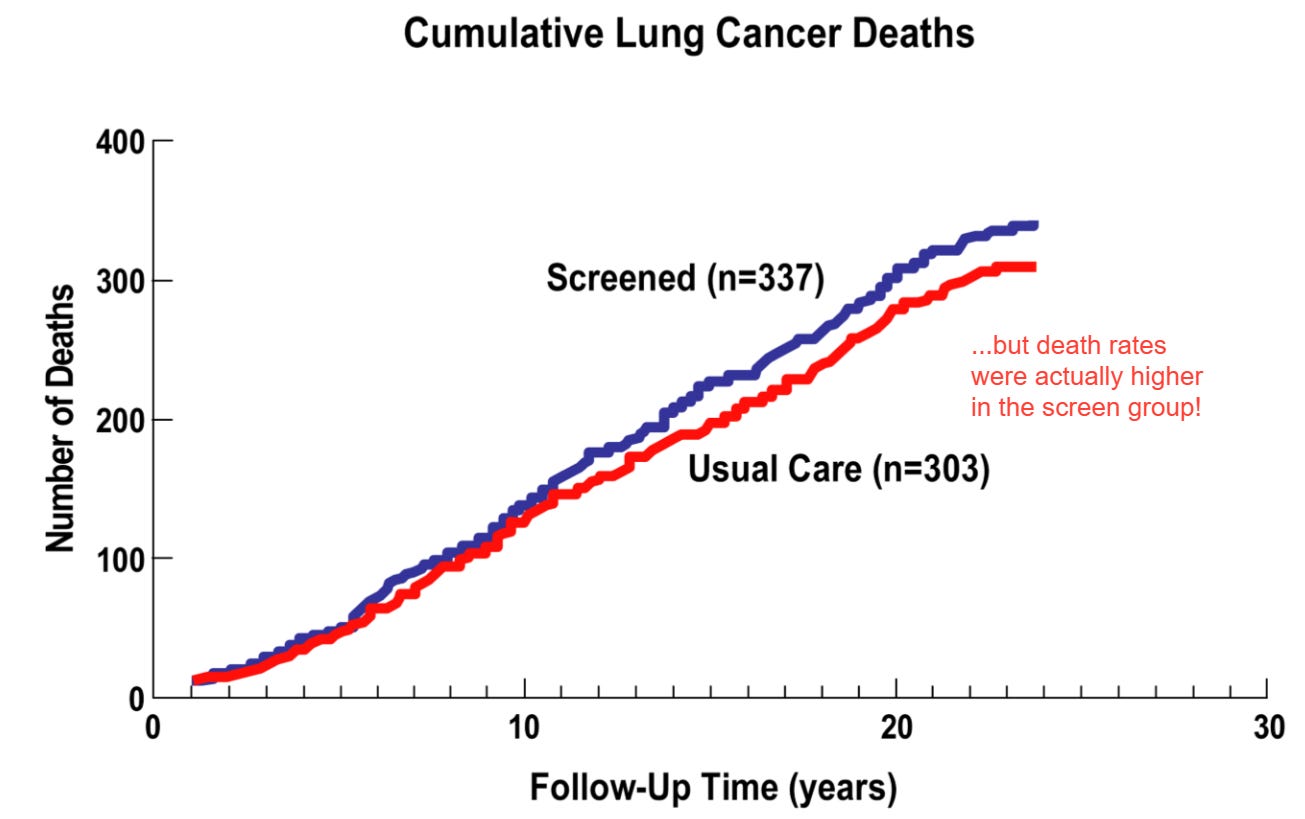
What matters, in many cases, is the death rate. Croswell et al cite the “…Mayo Lung Project, a randomized controlled trial of chest x-ray plus sputum cytology versus usual care for lung cancer screening, is one example. As [the following figure] shows, the 5-year survival rate after diagnosis appeared to strongly support the widespread implementation of screening: it was 36% among screened participants versus 19% for controls, or almost double. However, lung cancer mortality rates were not statistically significantly different between these two groups; in fact, they even trended towards an increase in deaths among those that were screened!”
For example, the ‘pre-cancerous’ condition cervical intraepithelial neoplasia (CIN 1) refers to abnormal cells affecting about one-third of the thickness of the cervical epithelium. It rarely becomes cancer and often goes away on its own. See Cervical Dysplasia: Causes, Symptoms, Diagnosis & Treatment.
Autopsy studies in men who have died of unrelated causes have shown subclinical prostate cancer in most elderly men. In other words, most elderly men have prostate cancer and die of something else. As for the living, 80% of prostate cancer diagnoses are in men whose cancer is confined to the prostate, with up to 99% still alive 10 years from diagnosis.