A lifetime of pain management, part 8
OxyContin: the sales pitch, the marketing, and the consequences!
In the previous post, I discussed the lack of evidence and guidelines to support the widespread use of opiates in chronic non-cancer pain (CNCP). In summary, as recently as 2018 there was still no evidence that opiates in CNCP meaningfully reduced pain or improved function. Furthermore, as we’ve known for centuries, long-term use of opiates causes side-effects, can paradoxically increase pain, and carries the risk of physical dependence and/or addiction.
Notwithstanding that lack of evidence, by the mid-90’s patients with CNCP were demanding opiates and doctors were prescribing them. While opiates had previously been reserved for acute pain and cancer pain, by 1999 CNCP accounted for 86% of the total opioid market in the US. Obviously, there was a huge shift in practice patterns.
Some argue that the shift was driven by economic considerations1. By 2001, OxyContin was the most frequently prescribed brand-name opiate in the United States.
“Having received FDA approval in 1995 for the management of chronic pain, the aggressive marketing campaign pursued by Purdue Pharma resulted in an increase in sales from $44 million and 316,000 prescriptions in 1996 to a combined total of nearly $3 billion and 14 million prescriptions in 2001 and 2002” (Source: OxyContin, prescription opioid abuse and economic medicalization)
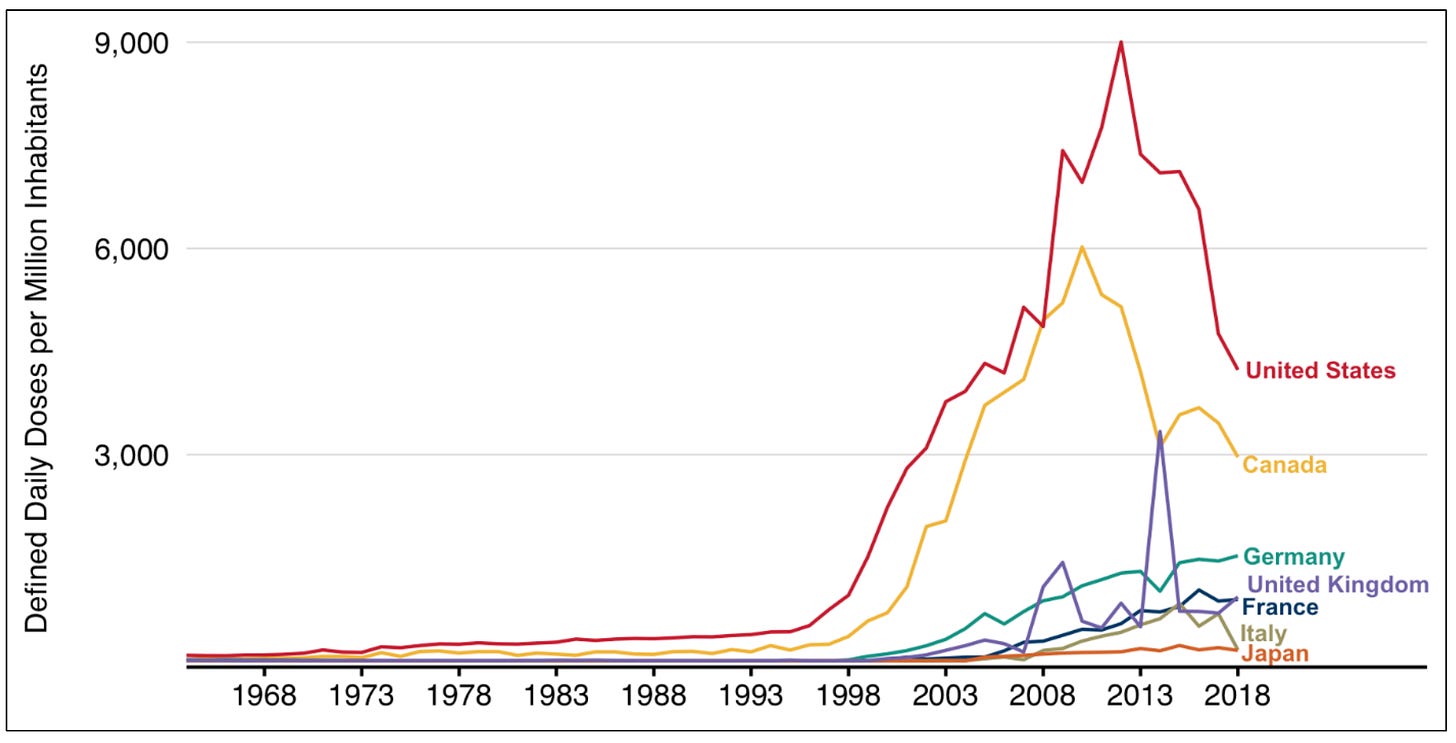
Similar growth was seen across Canada. For example, in Newfoundland, a 2004 OxyContin Task Force report contained the following data, showing enormous growth in the total quantity of OxyContin prescribed and a shift toward the higher strengths:
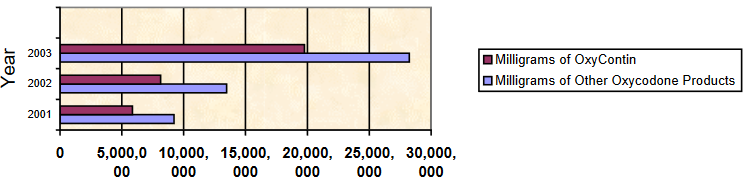
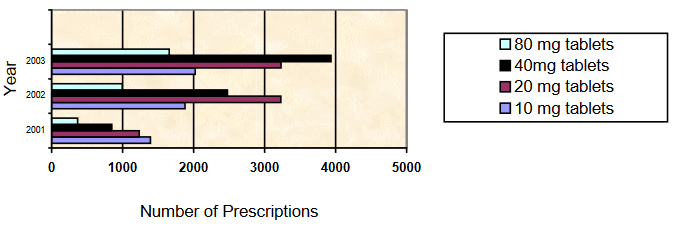
In short, the change in practice patterns involved a shift in the reasons for prescribing opiates, the specific choice of opiate, the number of prescriptions written, and the strength of the individual tablets.
This didn’t happen by accident. In this post, we’ll look at the role played by Big Pharma.
But first, a bit of history!
Back when I was a resident, the faculty were dead set against “drug reps”. We were told that they were obviously employed by Big Pharma for one reason, to influence our prescribing patterns. While free lunches and knickknacks seem harmless, they have an effect. Would Big Pharma really choose to spend money on something that didn’t work? Of course not!
“There is little doubt that detailers are effective in changing physicians' prescribing behaviour. In one study 25% of internal medicine faculty members and 32% of residents reported that they had changed their practice at least once in the preceding year based on a discussion with a detailer. In another study the more contact that Australian doctors had with detailers promoting temazepam the more quickly they began prescribing that drug and the more rapidly it became their hypnotic drug of choice. Physicians who requested formulary additions were much more likely than other doctors in the same hospital to have seen detailers from the manufacturers of the drugs in question.” (Source: Interactions between physicians and the pharmaceutical industry: What does the literature say?)
When I started in practice, I carried on with my aversion to drug reps. I was blown away by how many there were, and how persistent they could be in wanting to get through the door.
As my practice grew, I acquired partners who didn’t share my aversion, so the drug reps came to be a regular presence in the lunch room. On those occasions when I was desperate for a cup of coffee and absolutely couldn’t wait, I would run the gauntlet, trying to avoid eye contact, doing my best to look “pressed for time” while muttering “can’t talk now”. My strategy worked 90% of the time. The rest of the time I would get dragged into a “rote” presentation about some new drug with supposedly magical effects. My usual response was to specifically avoid prescribing whatever they were selling.
Based on the OxyContin story, I feel “vindicated”. Big Pharma is all about profit, not good patient care.
OxyContin, a new opiate!
In 1996, Purdue Pharma got approval to market their newly “invented” pain killer, OxyContin.
The active ingredient in OxyContin is oxycodone, an opiate drug first synthesized in 1916 and first used clinically in 1917. Chemically, it is very similar to codeine and hydrocodone.2
A different company, Merck, compounded oxycodone with scopolamine and ephedrine in 1928, labelling the combination as “SEE”, the "Miracle Drug of the 1930s", in Continental Europe and elsewhere. In World War 2, it was the German Army's preferred battlefield analgesic. Hitler’s doctor carefully recorded giving Adolf repeated doses of oxycodone, not that he had any battlefield injuries!
When I started practice in the 80’s, 5 milligrams of oxycodone could be found in combination with acetaminophen (trade name Percocet) or in combination with ASA (trade name Percodan). You could take one or two tabs at a time, so the single dose of oxycodone was 5-10mg. In my community (Dartmouth, Nova Scotia), we rarely prescribed it, preferring to use codeine in combination with acetaminophen (Tylenol #3, for example). The general thinking was that Percocet and Percodan were more addictive than Tylenol #3.3 Regardless, the fixed dose combination with acetaminophen or ASA meant that you would get toxicity from the acetaminophen or the ASA if you started taking more than the recommended dose, and there was no way to separate the opiate from the other ingredients.
And then, in the mid-90’s, Purdue Pharma came up with OxyContin. It wasn’t rocket science. They took that very old drug, oxycodone, slapped on their proprietary sustained-release pill coating (originally called “Continus” but later shortened to “Contin”), and thereby created something patentable.4 The timing was good, as the patents were about to expire on their similar drug, MSContin, a sustained-release version of morphine that they had marketed appropriately for use in palliative care.
Sustained-release is simply a system used in pills or capsules to make them dissolve slowly and release the active ingredient over a longer time, so that they can be taken less frequently while they keep more steady levels in the bloodstream.5 The hope is that patients will find it easier to remember to take their pills when they have to take them less often, and they’ll suffer less side-effects and withdrawal effects because the blood levels won’t go up and down so much. Of course, convenience comes at a cost, and some patients can’t afford the more expensive formulations.
For the drug company, besides the higher price, the other advantage of a sustained-release formulation is that it makes it difficult for your competitors to mimic the construction and behaviour of your product, meaning that prescribers and pharmacies will favor your trade name version, rather than switching to the cheaper generic version.6 In commercial words, your product is less price-sensitive, meaning you can charge a premium price without fear of losing your customers to the competition.
OxyContin is approved!
OxyContin was approved by the US Food and Drug Administration (FDA) late in 1995 and by Health Canada in 1996, based on application submissions provided by Purdue.
The subsequent history is well documented in the legal and medical literature. It’s widely accepted that there was “fraud” on behalf of Purdue and a “regulatory failure” by the FDA and Health Canada.
Leading up to its approval, there were no long-term studies of OxyContin’s effectiveness in chronic pain and no assessment of its addictive capabilities.
Even though chronic pain means persistent pain for over 3-6 months, and even though it was generally understood at the time that addiction occurs over periods of several days to weeks, the longest Purdue-sponsored OxyContin clinical trial lasted only 24 days, meaning that they offered no specific evidence of its long-term effectiveness in CNCP, and no way to assess the likelihood of addiction.
In essence, because opiates were known to work for pain in general, it was accepted that they would work for CNCP.
It was simply assumed that the risk of addiction was minimal due to the controlled-release formulation and the twice-daily dosing, which would avoid euphoria peaks and withdrawal troughs. The data for this was both flawed and misrepresented. First, there was no proof at the time that addiction was based on “peaks and troughs” and/or waves of euphoria. Next, they assumed patients would take it twice daily, so the addiction risk of dosing at more frequent intervals was not considered. Finally, they assumed that patients would swallow the pills whole. Unfortunately, if OxyContin was chewed, dissolved in water, or crushed the sustained-release coating was bypassed, releasing the entire oxycodone dosage at once.
In short, Purdue’s submission provided NO evidence in support of the indication for chronic non-cancer pain, nor did it include meaningful evidence pertaining to the risks of addiction and misuse. And yet OxyContin was approved, a mere 11 months after it was submitted to the FDA for review!7
OxyContin’s US product monograph stated that the drug was safe and effective for the treatment of “moderate pain”, with no stipulation that it be used only for acute pain or for end-of-life cancer pain. It claimed that misuse was “not a problem in patients with pain in whom [OxyContin] is indicated”. The underlying assumption was that patients taking OxyContin for the “right reasons” would take it in the “right way”. Nothing was mentioned about the risk of addiction (“misuse” being much less pejorative than “addiction”). Being designed to provide 12-hour pain relief, the monograph stated “if a breakthrough pain repeatedly occurs at the end of the dosing interval it is generally an indication for a dosage increase rather than more frequent administration”. No maximum dose was specified. The monograph cautioned against chewing, crushing, or dissolving the pill in water, which effectively told those inclined to misuse the medication exactly how to do it.
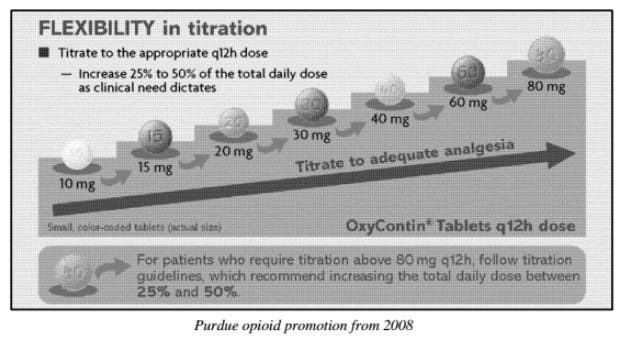
In Canada, the Health Canada-approved monograph likewise claimed that OxyContin was safe and effective for “chronic pain”, stating as well that “Drug abuse is not a problem in patients with pain in whom oxycodone [the active ingredient of OxyContin and the generic name of OxyContin] is appropriately indicated”. As in the US, the drug was marketed with no upper dose limit.
Following approval, on both sides of the border OxyContin was aggressively marketed as a long-acting opiate that could help patients with moderate to severe pain of all types, including CNCP. It’s a classic case study in the dirty tricks Big Pharma use to peddle their wares. 8
There were slight differences in terms of timing and approach in Canada and the USA, but overall things were pretty similar:
In the early 90’s, leading up to the release of OxyContin, Purdue fostered increasing interest in the treatment of pain, CNCP in particular, by contributing heavily to “independent” patient-advocacy groups such as the National Foundation for the Treatment of Pain and the Chronic Pain Association, and by sponsoring research that examined the effectiveness of opiates in CNCP.
These organizations played a major role in lessening the stigma associated with CNCP and its treatment.
Pharmaceutical companies, including Purdue, contributed heavily to the American Pain Foundation, the largest non-profit organization focusing on patients with pain. By 2000, the foundation received 60 percent of its funds in the form of unrestricted grants from pharmaceutical companies.9
To promote OxyContin, Purdue conducted all-expenses-paid pain management and speaker training conferences at 5-star resorts in Florida, Arizona, and California. These were attended by thousands of physicians, pharmacists, and nurses, some of which were then recruited and trained for Purdue’s national speaker bureau.
Key opinion leaders, including Dr. Russell Portenoy, Dr. Roman Jovey, and Dr. Brian Goldman10 promoted OxyContin at pain management seminars on both sides of the border, sponsored by Purdue.
While direct-to-consumer drug advertisements were not allowed in Canada, Canadians were exposed to direct-to-consumer OxyContin advertisements through US media.
Purdue funded over 20,000 pain-related educational programs through direct sponsorship or financial grants. This included multiple publications targeted at physicians (including those in training) and patients.
A pain management book, funded and copyrighted by Purdue, erroneously presented oxycodone as a weak opioid. For several years, that book was provided free of charge to doctors and health professions students at the University of Toronto, in support of their mandatory week-long pain management training.
Similarly, Purdue paid the Massachusetts General Hospital $3 million and proposed “areas where education in the field of pain is needed” as well as “curriculum which might meet such needs”.
Tufts University made a Purdue employee an adjunct associate professor in 2011. Purdue-written materials were approved for teaching Tufts students in 2014.
Through all those venues, Purdue aggressively promoted the use of OxyContin for use in CNCP, a much larger market than that for acute and cancer-related pain, and they specifically encouraged primary care physicians to adopt a more liberal approach to prescribing opioids, particularly the sustained-release forms. By 2003, nearly half the OxyContin prescriptions were written by primary care physicians.
With no evidence to support their claim, Purdue’s sales representatives, literature and audiotapes for physicians, brochures and videotapes for patients, and ‘‘Partners Against Pain’’ Web site all conveyed the message that the risk of addiction was “less than one percent”. One such pamphlet reassuringly said “Drug addiction means using a drug to get high rather than to relieve pain. You are taking opioid pain medication for medical purposes. The medical purposes are clear and the effects are beneficial, not harmful.”
“Partners Against Pain” was presented as an independent coalition of patients, doctors, and other health care providers dedicated to raising pain awareness. Ostensibly a grassroots organization, it was in fact created and funded by Purdue.11 Partners Against Pain promoted three messages: (1) pain was more prevalent than commonly believed, (2) pain could and should be treated more effectively, and (3) opiates should be part of that treatment12. Many of its pamphlets were written and signed off by Dr. Russell Portenoy, whose clinics received millions of dollars in donations from Purdue.
Purdue more than doubled its US sales force and total physician call list. A generous bonus system13 encouraged sales representatives to increase sales of OxyContin in their territories, including a multifaceted campaign specifically aimed at physicians with high rates of opioid prescriptions. Indeed, they already had existing data showing which doctors were more inclined to prescribe their previous opiate, MS Contin.
The marketing campaign targeted specific groups who would not normally have been considered candidates for opiates, including the elderly, veterans, opiate-naive patients, and patients with chronic conditions like osteoarthritis.
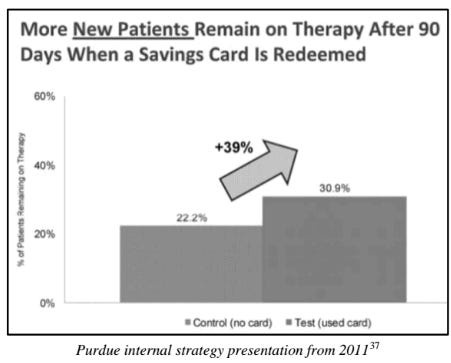
To get patients started, in the US Purdue offered coupons for a free limited-time prescription for a 7-to-30-day supply. Patients could get their first batch of OxyContin for free! What could possibly go wrong? When the program was ended in 2001, approximately 34,000 coupons had been redeemed.
The underlying message was that “OxyContin was the opiate to start with and to stay with.”
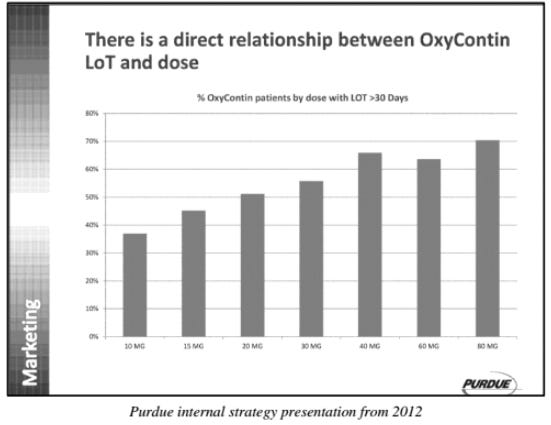
Purdue promoted the concept of “pseudo-addiction”, claiming that when patients started to seek higher doses of opiates, “watch the clock”, supplement with illegal opiates, or resort to deceptive behaviour (i.e. “double doctoring”)14, it was a sign that their pain was under-treated, for which the “appropriate” response was an increase in the dosage prescribed.
OxyContin, unsafe at any dose!
OxyContin was a massive commercial success. Marketing works!
Unfortunately, by 2004, OxyContin was also a leading drug of abuse in North America.15 Drug abusers discovered almost immediately that they could simply crush the controlled-release tablet and swallow, inhale, or inject the powder for an intense high. This should not have come as a surprise. Purdue’s previous sustained-release morphine product, MS Contin, had been similarly abused in the late 1980s, and Purdue had demonstrated in 1995 that 68% of the oxycodone could be extracted from an OxyContin tablet simply by crushing the pill.
The problems with OxyContin were slow to attract the attention of the authorities. A 2003 FDA Warning to Purdue Pharma stated “Your journal advertisements omit and minimize the serious safety risks associated with OxyContin, and promote it for uses beyond which have been proven safe and effective... your journal advertisements fail to present in the body of the advertisement critical information regarding limitations on the indicated use of OxyContin, thereby promoting OxyContin for a much broader range of patients with pain than are appropriate for the drug”. Even so, the regulators did not come down hard on Purdue. Some advertisements were retracted, and the package insert was updated, but OxyContin was still FDA approved to treat CNCP.
At the same time. the FDA also warned physicians about the risks and encouraging them to be more cautious when prescribing OxyContin.
While the FDA’s actions were known to Health Canada, no similar actions were taken in Canada, perhaps because OxyContin abuse was mistakenly thought to be a uniquely American problem. It was 2006, 5 years after the FDA’s label revision and Black Box Warning, when Health Canada revised the drug’s monograph by stating the danger of breaking, chewing or crushing the pills. They also slightly changed the misleading sentence regarding the abuse liability of the drug, adding the word “usually”, so it said “Drug abuse is usually not a problem ...”.
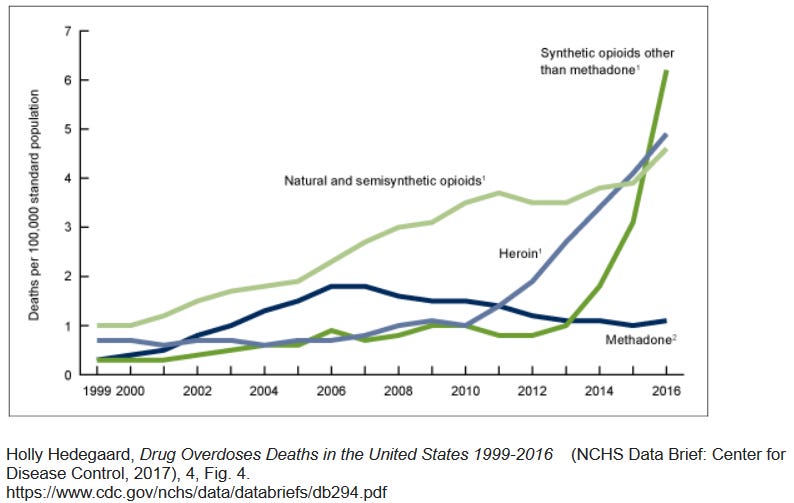
OxyContin was reformulated in 2012 as the crush-, chew-, injection- and dissolve-resistant OxyNEO, causing some users to switch to heroin. Doctors became increasingly hesitant to prescribe opiates, so many patients who were legally dependent on OxyContin were cut off from their supply, resorting to black market opiates such as heroin and fentanyl.
After 2010, illegal opiates became the driving force in the opiate crisis. Because they are much easier to overdose on, overdose deaths increased.
In 2022-23, Purdue Pharma reached a settlement with eight US states plus the District of Columbia, declaring bankruptcy. In response to Purdue’s problems, their competitors increased their marketing efforts, even in counties where the opiate crisis was known to be severe. Business is business, after all!
This process is termed “economic medicalization”, where a condition previously NOT treated with medication is magically transformed into a condition “requiring” medication, the motivation being corporate profits. Pain experts, however, described the change as “correcting” the historical undertreatment of patients with severe chronic pain. Corporate greed or societal good? Either way, “economic medicalization involves a sharp ethical divergence between the goal of [Shareholder Wealth Maximization], associated with business ethics, and the norms of science and the scientific method, associated with medical ethics.” (Poitras, 2012)
Hydrocodone is the active ingredient in Vicodin, the preferred drug of Dr. Gregory House.
Oxycodone sits on Schedule II of the USA’s Controlled Substances Act, meaning that it has a high potential for abuse, with use potentially leading to severe psychological or physical dependence. Schedule II drugs are also considered dangerous.
Products containing less than 90 milligrams of codeine per dosage unit (including Tylenol #3) are Schedule III drugs, with a moderate to low potential for physical and psychological dependence.
So, we were justified in thinking that Tylenol #3 was safer than Percocet or Percodan.
Schedule I is reserved for drugs with no currently accepted medical use and a high potential for abuse.
I believe the best phrase to describe this is “putting lipstick on a pig”, meaning “making superficial or cosmetic changes to a product in a futile effort to disguise its fundamental failings”.
There are many ways to accomplish this trick.
Some jurisdictions have legislation that requires pharmacies to dispense the lower cost generic, if there is one, assuming the generic is “bioequivalent”. It’s harder to prove bioequivalence with sustained-release formulations, as you are dealing not just with the quantity of the active ingredient, but also how it is released from the pill or capsule over time.
This process usually takes years! There’s some evidence that somebody who worked for the FDA took a bit of “time off” to write up Purdue’s submission to the FDA, for which he was later rewarded with a much better paid job at Purdue.
One of the owners of Purdue, Arthur Sackler, got his start in advertising, where his greatest success was marketing Valium and Librium to anxious women in the 60’s and 70’s, all the while ignoring the risk of addiction!
The grants may be “unrestricted”, but you can guarantee they’ll dry up if the funder doesn’t like what the organization is doing.
Dr. Goldman “fessed up” in an editorial published in The Globe and Mail on March 23, 2012. His apology was “mixed”, in that he claimed to be “proud of the role I played in making opioids more available to patients who need them by teaching doctors how to prescribe them carefully”, while also saying “I no longer believe it to be possible for educational courses paid for by drug companies to be free of corporate bias”.
This is called “Astroturfing”. Astroturf looks like grass, but it isn’t. Industry-funded patient advocacy groups look like grassroots organizations, but they aren’t.
These messages were “generic” and did not mention OxyContin specifically. Purdue created a larger market for its products simply by promoting the concept of pain relief. ads of this sort continue (i.e. “Ask your doctor if there’s a better way to treat your [insert condition name here]”).
According to this article: “In 2001, in addition to the average sales representative’s annual salary of $55000, annual bonuses averaged $71500, with a range of $15000 to nearly $240 000. Purdue paid $40 million in sales incentive bonuses to its sales representatives that year.”
Internal memos show that some doctors on the Purdue payroll recognized that these were clear signs of actual addiction. Their concerns were ignored.
Put another way, not only did Purdue take over the prescribed opiate market, they made a big dent in the illegal drug trade as well! Business is business, after all.




Rick - another great read! Thanks.
A few stories/comments.
I had a lady from NB once, travelling to Nfld, who ended up in our ER in CB in severe narcotic withdrawal. She was on 800mg of morphine per day for chronic pain ("fibromyalgia"). Her doctor had assured her that she would never get addicted because "she had real pain" and wasn't using it for a high like addicts. She forgot her pills at home, and got past Truro before she realized. Because (thank God!) she wasn't addicted, she didn't have to go home to get them.
RE: drug rep visits. Very quickly after starting practice (within a few months), drug rep visits went from several times per week down to almost zero. I later found out through a pharmacist friend what I've since had confirmed (and you talk about). Big Pharma buys your prescribing history from pharmacies. They can very quickly find out who prescribes cheap, generic, old-fashioned, well-studied tried-and-true drugs, and who prescribes the expensive glittery newest things that have huge profit margins for Pharma. They target their resources on the "soft targets".
I read carefully about Vioxx and didn't prescribe it, even though it was pushed HARD. I remember one drug rep vaguely threatening that, since it was safer on the stomach as they claimed, if one of my patients on an old NSAID had a GI bleed, I would be legally liable for not having prescribed Vioxx.
You mention Hitler's drug habit. There is a great book I read recently called "Blitzed" about the ubiquitous use of drugs in the Third Reich, including stimulants and narcotics. It was the start of transhumanism - the idea that we can transcend nature and become super-beings.
The other thing that really resonates with me is the complicit evil of people like Brian Goldman. He is a guy who, even when I agree with his point, manages to come across as a condescending asshole. This sounds catty, but I know someone who worked closely with him in ER in Toronto and said he was a terrible doc. Slow, ponderous, and very prone to over-testing, overdiagnosis, over-treatment and over-referral. And yet for years on White Coat Black Art he presents himself as the wise one - all-knowing, all-seeing, sitting on his perch looking down on the rest of us mere mortal physicians. When I found out years ago that he had been a pharma shill for Oxycontin, it was no surprise to me. He loves to hear himself talk, loves to be the expert, and is overly credulous. The perfect person to swallow the pharma bunk and then regurgitate it.
I suppose the various Drug User Liberation Fronts may also be Astroturf organizations.